CLSI Blog Articles
Read the latest articles about CLSI and laboratory standards in the official CLSI Blog. Browse our most recent blog articles below.
World Health Day: Celebrating 75 Years of Health Advocacy
On April 7th, we commemorate World Health Day, which marks the 75th anniversary of the establishment of the World Health Organization (WHO), in 1948. This year’s theme, “My Health, My Right,” emphasizes the fundamental right of every individual to access quality health services, education, and information. It also underscores the importance of safe drinking water, clean air, good nutrition, quality housing, decent working and environmental conditions, and freedom from discrimination.
Read MoreA Week for Collaboration and Consensus: CLSI’s January 2024 Meetings
CLSI’s Committees Weeks are an opportunity for the CLSI expert community to meet and connect with colleagues for a week of information sharing, consensus building, networking, and camaraderie. Committees Weeks are where the hard work behind CLSI’s standards and guidelines takes place—committees and subcommittees, following CLSI’s voluntary consensus process, work together on new and revised CLSI standards in development.
Read MoreAST News Update January 2024: Acinetobacter – the Bad, the Awful, and the Downright Ugly
Between 2018-2021, the National Healthcare Safety Network (NHSN) reported that 0.4% (n=1,951) of hospital-acquired infections (HAIs) in the United States were caused by Acinetobacter spp. Of these, 28-45% were not susceptible to carbapenem antibiotics (ie, intermediate or resistant).1 CDC’s 2019 Antibiotic Resistance Threats Report estimated that there were 8,500 carbapenem-resistant Acinetobacter cases in hospitalized patients in 2017.2 Consistently, the A. calcoaceticus-A. baumannii complex (A. baumannii) is the largest cause of clinical Acinetobacter spp. infections and is most often recovered from respiratory specimens.
Read MoreAST News Update January 2024: Antifungal Body Site Reporting for Candida spp.
A 59-year-old woman with metastatic breast cancer was admitted to a hospital for fever and suspected sepsis 12 days after chemotherapy-induced neutropenia (<1,000 neutrophils/μL in peripheral blood; normal range 2500-7000 neutrophils/μL). The patient was started empirically on cefepime, vancomycin, and anidulafungin. Two sets of blood cultures grew Candida tropicalis on day 3 post admission. Since this isolate was recovered from the blood, both species-level identification and susceptibility testing were performed. At the time of blood culture positivity, the patient did not report any visual symptoms, and dilated fundoscopy of the eyes on day 4 did not reveal any signs of ocular involvement.
Read MoreAST News Update January 2024: Practical Tips for Using Newly Formatted Tables 1 in CLSI M100 33rd Edition
In 1972, CLSI, formerly known as the National Committee for Clinical Laboratory Standards (NCCLS), published “Table 1” in one of the earliest NCCLS AST documents. Table 1 was intended to help clinical laboratories decide which antimicrobial agents to test and report on specific bacteria. New drugs and new comments were added to the Tables 1 over the ensuing decades, but the format for these tables did not change.
Read MoreAST News Update January 2024: Sulbactam-Durlobactam: The “double beta-lactamase inhibitor” drug
Many laboratorians are familiar with sulbactam, a β-lactamase inhibitor that is commercially combined with ampicillin, which was introduced in the US in 1986 to make ampicillin-sulbactam (“Unasyn”). Like many older β-lactamase inhibitors, sulbactam is itself a β-lactam, but has poor antimicrobial activity against most bacteria.
Read MoreNew Training Course for Latin American and Caribbean Veterinary Labs Results in Stronger Standards, Safer Food
CLSI recently partnered with the Ohio State University (Ohio State) and the Inter-American Institute for Cooperation on Agriculture (IICA) for the first training of its kind that brought together official veterinary diagnosticians in Latin America and the Caribbean (LAC) working in national reference labs monitoring antimicrobial resistance (AMR).
Read MoreCLSI Celebrates One Health Day 2023
November 3, 2023 is the eighth annual One Health Day. This global campaign highlights the need for an integrated approach to tackle shared health threats around the world. The One Health approach spans a multitude of disciplines and sectors to achieve optimal health outcomes for people, animals, plants, and their shared ecosystems, through coordinated interventions.
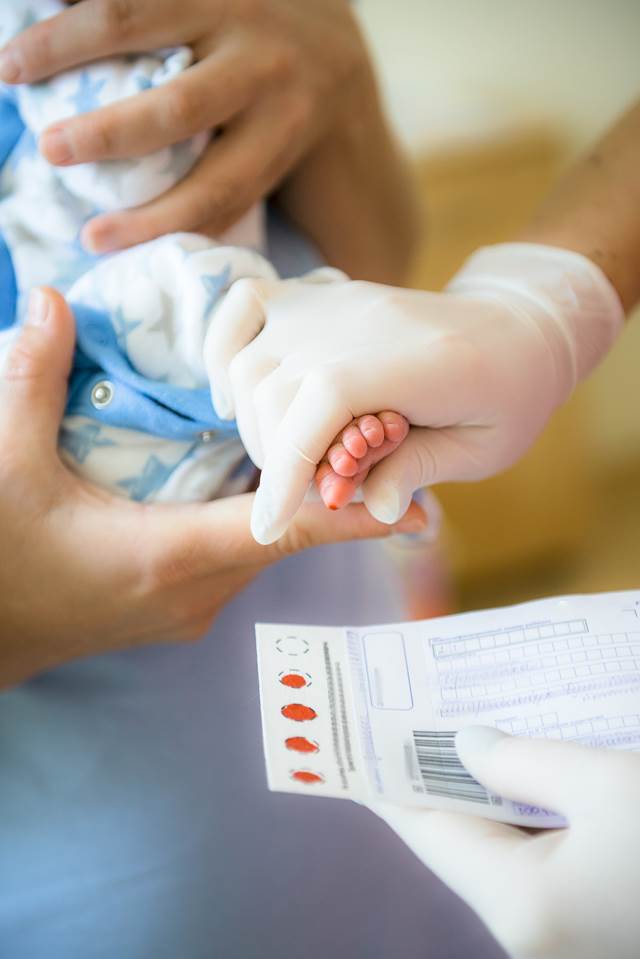
Read MoreRecent Technologies for Newborn Screening
Newborn screening (NBS) is an important component of public health that helps health care providers identify hereditary and metabolic disorders in newborns and enables early therapeutic intervention, which can be lifesaving or altering. Newborn screening programs are typically state- or country-based public health programs. Today, newer and more complex technology is increasingly being integrated into NBS.
Read More